NGI and NOAA's AOML Study Reveals River Runoff Creates a Buffer Zone for Ocean Acidification in the Gulf of Mexico
By AOML Communications

The pH scale indicates the concentration of hydrogen ions in a solution. It works in an inverse way such that the higher the concentration of hydrogen ions, the more acidic and the lower the pH value will be. The lower end of the scale is acidic, and the higher end of the scale is basic or alkaline. Photo by NOAA Pacific Marine Environmental Laboratory
The research, published in Geophysical Research Letters, used models to identify the main drivers of ocean acidification for different regions of the gulf. They provide evidence that river alkalinity has counteracted the progression of ocean acidification for coastal areas along the gulf.
Ocean acidification refers to a reduction in seawater pH over time, mainly caused by increased levels of carbon dioxide in the atmosphere being absorbed into the ocean. Seawater chemically reacts with carbon dioxide to form carbonic acid, causing the ocean to become more acidic.
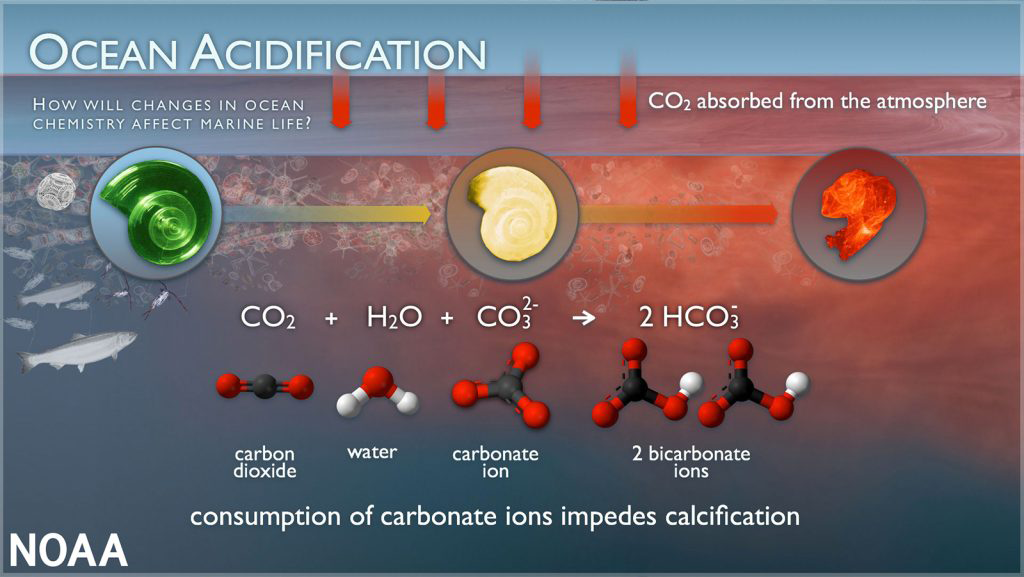
This image shows a pteropod shell dissolving over time in seawater with a lower pH. When carbon dioxide is absorbed by the ocean from the atmosphere, the chemistry of the seawater is altered and conditions become more acidic. Photo by NOAA
Corals and shellfish need a higher aragonite saturation state and less acidic waters, i.e., higher on the pH scale, to thrive. If waters become too acidic, less coral reef habitat will be available for fish and other reef-dwelling animals, diminishing biodiversity and marine ecosystem health.
The Mississippi River has a relatively high level of alkalinity for a freshwater body. Over recent decades, agricultural practices such as liming (adding neutralizing materials to lower soil acidity) and water quality improvements have contributed to the increased water alkalinity in the Mississippi River system. Alkalinity acts as a neutralizing factor to make a solution less acidic and more basic or alkaline.
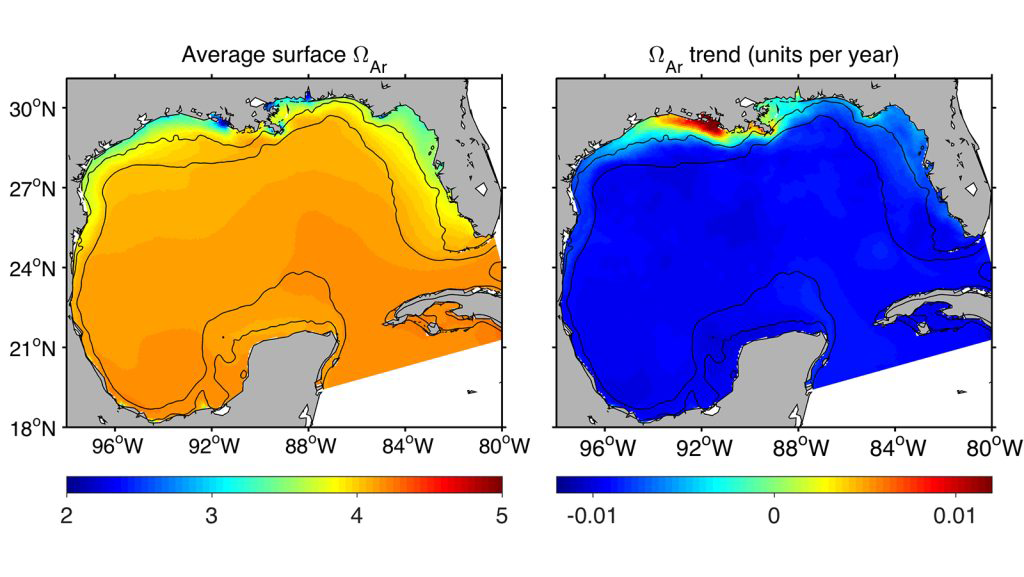
Average surface aragonite saturation state (ΩAr), an indicator for ocean acidification (left), and its long-term trend (right) showing less acidic waters (with a higher aragonite saturation state) around the northern inner-shelf of the Gulf of Mexico. This ocean acidification buffer was caused by the Mississippi-Atchafalaya River's alkaline water mixing and neutralizing the seawater in that area. Graphics by Fabian Gomez
Ocean acidification is a major environmental stress factor that contributes to the degradation of valuable marine resources in the gulf. A recent report by NOAA's Office of Coastal Management highlighted the economic value of the Gulf of Mexico's coastal and marine ecosystems: they support approximately 598,000 workers and are the largest contributor to America's blue economy, with an estimated $104 billion dollars coming from oil and gas production, marine shipping, and the fishing industry.
NOAA's research to better understand ocean acidification will help preserve and manage coral reefs and other marine species that gulf coastal communities depend on for fishing, tourism, and other economic drivers in the region.
- NGI Supports Going into the Eye of the Storm for Better Hurricane Predictions
- MSU Sets New Record for Research Expenditures, Climbs to No. 88 Nationally in Latest NSF Rankings
- Geosystems Research Institute Scientist Advancing Global Science with Remote Sensing and Imaging Analysis
- MSU's Center for Cyber Innovation is Part of Mississippi Cyber Security Initiative
- The Center for Advanced Vehicular Systems (CAVS) Defines the Future of Autonomous Vehicles with Quantum R-Series Edge Storage
- GRI Researcher and Geosciences Professor Awarded for Work in Geoscience Education by National Teacher Organization
- CCI Researcher's Inspirational Story Featured on Mississippi State's, 'Our People'
- MSU NGI Researcher and Geosciences Faculty Member Named President of National Weather Association
- MSU's Jordan Named to Oak Ridge Associated Universities Board of Directors
- Renowned Italian Scientist Visits MSU's Geosystems Research Institute for Fellowship in Precision Agriculture
- MSU's CCI Helps Mississippi Educators Train Soldiers
- MSU's CCI and Department of CSE Selected to Join U.S. Cyber Command Academic Engagement Network
- NGI Researcher, Rudzin, Earns Postdoctoral Award for Atmospheric-oceanic Hurricane Research
- GRI Researcher Renee Clary Honored as a New Fellow of the Mississippi Academy of Sciences
- NGI Supported Study Finds Existing and Planted Shoreline Vegetation Respond Differently to Breakwater Protection
- MSU, ERDC Celebrate Successful Partnerships Promoting Cutting-edge, Collaborative Research
- GRI Researchers, Partnership Middle School Install Interactive Weather station, Help Students Become 'Citizen Scientists'

